top of page

Recent Posts






All Posts


Is Expiry Labelling Mandatory in Japan?
Background In Japan, cosmetics are primarily governed by the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices(Act No. 145 of 1960) (also known as the Pharmaceuticals and Medical Devices Act (PMDA), formerly known as the Pharmaceutical Affairs Act (PAA) or the 薬事法 (Yakujihō). In accordance with the Article 61 of PMDA, cosmetics must have the following information labelled on their immediate container or immediate packagin
51 minutes ago


Ministry of Food and Drug Safety (MFDS) and Rural Development Administration (RDA) List Migratory Locust as 10th Edible Insect
The partial amendment to the Korean Food Code (No. 2026-40), published by the Korean Ministry of Food and Drug Safety (MFDS) on 19 May 2026, represents a significant regulatory development for Korea’s novel food and edible insect industry. Through this amendment, migratory locust (Locusta migratoria), locally referred to as “풀무치 (Pulmuchi),” was officially reclassified from a temporarily recognised food ingredient to a general food ingredient officially listed in the Korean F
2 days ago


Japan Launches Public Consultation to Establish the OELs for 48 Subtances
Background Following amendments to the Enforcement Order of the Industrial Safety and Health Act (ISHA), effective 1 April 2023, employers are required to take measures to minimize workers' exposure to hazardous chemical substances that are subject to risk assessment obligations. To reduce occupational exposure risks, employers must implement control measures in accordance with the hierarchy of controls, prioritizing the following approaches: Substitution of hazardous substan
2 days ago

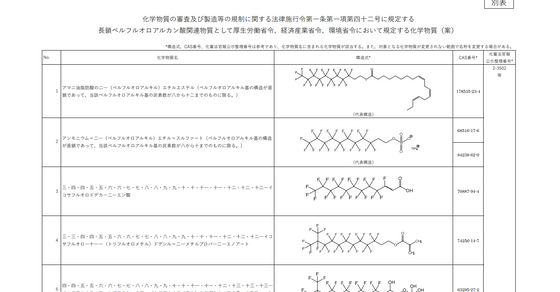
JAPAN - 156 Long-chain Perfluoroalkanoic acid (LC-PFCA) Related Substances, to be Regulated as Class I Specified Chemical Substances
Background At the twelfth meeting of the Conference of the Parties to the Stockholm Convention on Persistent Organic Pollutants (SC COP-12), held from 28 April to 9 May 2025, Parties adopted amendments to Annex A of the Convention to list three additional persistent organic pollutants (POPs): the pesticide chlorpyrifos and two groups of industrial chemicals, namely long-chain perfluorocarboxylic acids (LC-PFCAs) and related substances, and medium-chain chlorinated paraffins (
3 days ago


Japan Expands PFAS and POPs Controls Under CSCA: PFHxS-Related Substances, LC-PFCAs, MCCPs and Chlorpyrifos Designated as Class I Specified Chemical Substances
Japan continues to strengthen its chemical management framework under the Chemical Substances Control Act (CSCA), introducing significant new restrictions on per- and polyfluoroalkyl substances (PFAS) and Persistent Organic Pollutants (POPs) in 2026. The latest amendments expand the scope of substances designated as Class I Specified Chemical Substances (Class I SCS)—the most stringent regulatory category under the CSCA—resulting in broad prohibitions on the manufacture, impo
5 days ago


South Korea’s Cosmetics Act Amendment No. 21709: AI-Generated Advertisements May Now be Deemed as Unfair Advertising
On 26 May 2026, South Korea promulgated Amendment No. 21709 to the Korean Cosmetics Act, signaling a major regulatory shift in how cosmetics advertising will be assessed in the era of generative artificial intelligence (AI). The amendment will enter into force on 27 November 2026. The amendment reflects growing concerns among regulators regarding the rapid proliferation of AI-generated promotional content, including synthetic influencers, deepfake endorsements, fabricated tes
May 26


Korea|Partial Amendment of the Korean Food Code (2026-40)
The Korean Ministry of Food and Drug Safety (MFDS) has published a partial amendment to the Korean Food Code (FC) (No. 2026-40) on May 19, 2026, introducing significant revisions aimed at strengthening public health protection, improving regulatory consistency, and reducing import and export trade friction associated with microbiological compliance verification. The amendment seeks to reinforce the scientific basis of food safety management by: introducing statistical concept
May 25


Partial Amendment of Specifications And Standards for Foods, Food additives, etc.
The Consumers Affairs Agency (CAA) has published on 20 May 2026 (under Cabinet office Notification No. 71 of 2026 ) a partial amendment of Specifications and Standards for Foods, Food additives etc. (Act No. 370 of 1959) to revise the maximum residue limits (MRL) of Isocycloseram, Oxolinic acid, Broflanilide, Quizalofop-ethyl, Quizalofop-tefuryl, Pyflubumide, Phenmedipham and Benzyladenine based on Article 13, Paragraph 1 of the Food Sanitation Act (Act No. 233 of 1947). Main
May 21


Partial Amendment of Specifications And Standards for Foods, Food additives, etc.
The Consumers Affairs Agency (CAA) has published on 19 February 2026 (under Cabinet Office Notification No. 7 of 2025) a partial amendment of Specifications and Standards for Foods, Food additives etc. (Act No. 370 of 1959) to revise the maximum residue limits (MRL) of Isotianil, Clofentezine, Validamycin and Broflanilide based on Article 13, Paragraph 1 of the Food Sanitation Act (Act No. 233 of 1947). Additionaly, Acetylcysteine (feed additive) has been added to the list of
Feb 23


Korea|Public Consultation on (Draft) Partial Amendment. of Standards and Specifications for Food Utensils, Containers and Packages (UCP)(No.2026-009)
South Korea's Ministry of Food and Drug Safety (MFDS) has launched a public consultation on 8 January proposing a partial amendment (No.2026-009) to the "Standards and Specifications for Utensils, Containers, and Packaging (UCP)" The draft proposes to expand the scope of physical synthetic resin recycled raw materials* used in the manufacture of utensils, containers, and packaging (UCP) to include polypropylene (PP) in addition to polyethylene terephthalate (PET). In order t
Jan 13
bottom of page


